EPA Disposal Guidelines in Iowa are now different regarding hazardous pharmaceutical waste. Don’t worry. It’s a good thing – and a better fit for healthcare. It provides regulatory relief for hospitals and other large quantity generators of hazardous waste pharmaceuticals.
Part of this sweeping legislation is 40 CFR 266 Subpart P. Below is a recap of this federal ruling, who needs to comply, and how.
Iowa First to Adopt New EPA Rules
August 21, 2019 is the date the new EPA rules apply in Iowa, Alaska, the US Territories, and the Indian Country. If you’re unsure of whether or not your facility is a “healthcare facility” under this new rule, please consult with your local regulator. All other states have some leeway (until July 2021) when they adopt these new federal regulations.
This chart gives an idea of the timing when it may take place, such as in Wisconsin and Illinois. Note that some states’ current regulations may actually be even more stringent than the new EPA laws, and will follow those instead.
That said, there is one aspect of Subpart P that every state must follow as of August 21, 2019 — the nationwide Sewer Ban, which we’ll discuss in more detail below. Iowa healthcare facilities must prepare for the Sewer Ban and a few other changes. If you work for or manage one of these facilities in Iowa, it’s important to comply to these new regulations. For all others, follow your state’s existing regulations until these new rules become a state law.
Does My Iowa Healthcare Facility Need to Comply to Subpart P?
Healthcare facilities often fall into three buckets:
- If you provide preventative, diagnostic, therapeutic, rehabilitative, maintenance or palliative care to humans or animals.
- Your facility provides counseling services, assessments or procedures, with respect to patients’ physical, mental condition, or functional status.
- You distribute, sell, or dispense pharmaceuticals, including over-the-counter pharmaceuticals, dietary supplements, homeopathic drugs, or prescription pharmaceuticals.
With that in mind, here are some examples of healthcare facilities who might need to make changes because of this rule:
- Hospitals
- Health Clinics
- Psychiatric and Substance Abuse Centers
- Ambulatory Services and Ambulatory Surgical Centers
- Physicians’ Offices
- Optical and Dental Providers
- Chiropractors
- Veterinary Clinics and Hospitals
- Long-Term Care Facilities (administer pharmaceuticals to one or more individuals there)
- Pharmacies (including Mail-Order and Long-Term Care)
- Retailers of Pharmaceuticals
- Wholesale Distributors
- Third Party Logistics Providers that serve as forward distributors
New EPA Disposal Guidelines Will Affect These Facilities
Ultimately, this rule will affect these three types of healthcare facilities that regularly accrue hazardous waste pharmaceuticals. Namely:
- Large Quantity Generators (LQG): Generate more than 2,200 lbs. of hazardous waste per month.
- Small Quantity Generators (SQG): Generate more than 220 lbs. of hazardous waste but less than 2,200 lbs. per month.
- Reverse Distributors: Those who receive or take back prescription pharmaceuticals from another organization to verify a manufacturer’s credit
Very Small Quantity Generators (VSQGs), or those who generate less than 220 lbs. of hazardous waste pharmaceuticals per month, are not beholden to Subpart P, (except for the Sewer Ban). They can continue to follow their previous way of handling their pharmaceuticals and acutely hazardous P waste. However, they may opt into Subpart P. Or, they can also send their hazardous waste pharmaceuticals off to an affiliate site that operates under Part 266 Subpart P.
What is a “Hazardous Pharmaceutical Waste?”
A review of the formulary list from the pharmacy will help you determine what is a hazardous pharmaceutical waste — and what isn’t. Your pharmacist will know – and may likely even tell you that a majority of what’s on the shelf is actually non-hazardous. The whole point of Subpart P is to make sure you properly dispose of pharmaceutical hazardous waste. Most importantly, not flush it down the sewer, where it can pollute the waterways.
Commingling of hazardous and non-hazardous waste pharmaceuticals in the same container is acceptable. But remember then, the whole container is now hazardous waste.
Changes to Comply with Subpart P in Iowa
We’ve discussed who this might affect, now let’s dive into the nuts and bolts of the upcoming changes. Here are six big new changes from what you might or might not be doing that are in Subpart P and the new Nicotine Amendment
1. Sewer Ban – 266.505
In the past, many healthcare facilities flushed hazardous waste pharmaceuticals down the sink or in the toilet. This practice will soon be met with harsh penalties. In short, all states will no longer be able to sewer hazardous pharmaceutical waste as of August 21, 2019.
If you’re unsure of where to start, begin by obtaining a formulary list from your designated pharmacist or pharmaceutical provider to make you aware of what hazardous pharmaceutical waste you have on site.
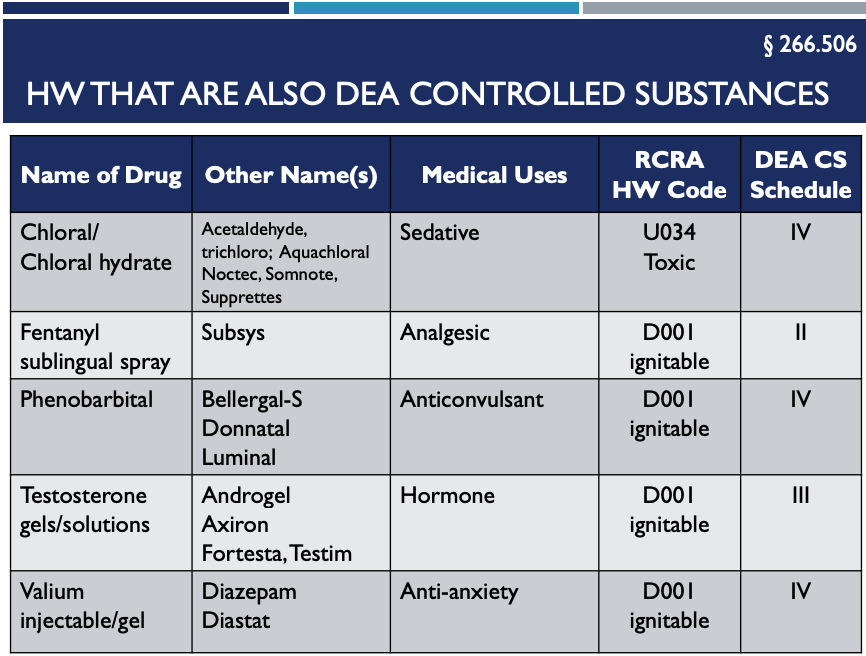
2. Controlled Substances – 266.506
Here are five controlled substances that are also RCRA Hazardous Waste Pharmaceuticals. They must not be sewered:
The five DEA hazardous substances noted above are EXEMPT from RCRA if they are:
- Not flushed down the drain,
- Managed in compliance with DEA Regulations,
- Destroyed by a method that the DEA has deemed in writing to meet their non-retriable standards, or
- Thermally destroyed at a permitted hazardous waste disposal facility.
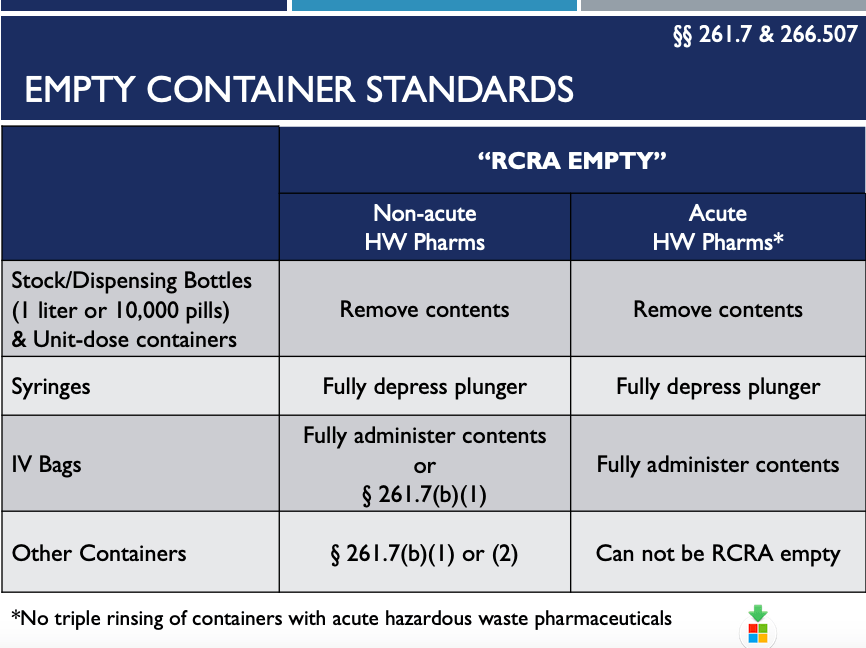
3. “RCRA Empty” – 261.7 & 266.507
The new 266 Subpart P Regulations cover other areas, including defining “RCRA Empty”, as shown in the chart below.
As noted in the above chart, “RCRA Empty” means fully remove medicine from dispensing bottles. For syringes, fully depress the plunger. And, for IV bags, fully administer contents before disposing of it. Basically, once you’ve removed everything from it by pouring, pumping, and aspirating the medicine, it is “RCRA Empty”. Residues remaining inside of a container will no longer be regulated as hazardous waste pharmaceuticals.
What does this “RCRA EMPTY” change mean for you? In the past, many healthcare facilities triple rinsed containers when there was still a trace amount of hazardous waste pharmaceuticals in them. No more. As of August 21, 2019, triple rinsing of RCRA pharmaceutical empty containers with acute hazardous pharmaceutical waste is not required/allowed.
4. Nicotine Disposal Amendment
The nicotine disposal amendment has been adopted by Iowa. It is effective August 21, 2019. Other states have to adopt this amendment. Over the counter nicotine items such as nicotine gum, patches, wrappers and lozenges are no longer considered acutely hazardous (P075) waste. They will now be considered non-hazardous waste.
Keep in mind that nicotine in its purest form will still be acutely hazardous and must be disposed of as such. This includes unused formulations of nicotine that can be found inside e-cigarettes, cartridges and vials.
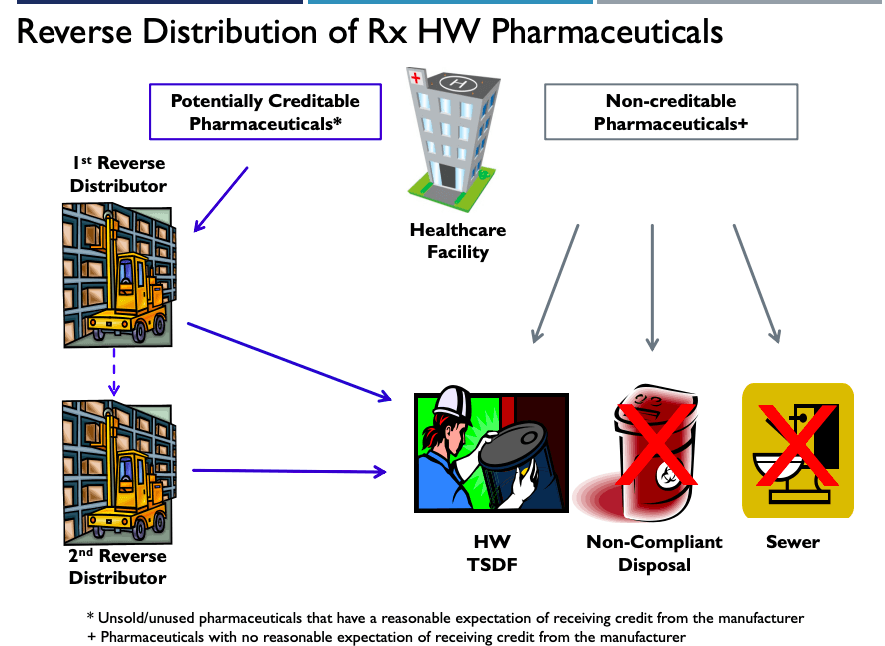
5. Reverse Distribution – 266.500
In years past, healthcare facilities have typically given most or all of their pharmaceutical waste to Reverse Distributors. This was regardless of whether they thought they could get credit for their expired medication. Subpart P changes things.
The EPA now says healthcare facilities can only give Reverse Distributors “potentially creditable” pharmaceutical waste. Furthermore, pharmaceutical waste must be identified on site as potentially creditable or non-creditable pharmaceutical waste, and not hauled off site and identified elsewhere. Finally, it must be evaluated within 30 days to determine its creditability.
Below are a couple lists to help you determine what waste is potentially creditable.
- Potentially Creditable Waste:
- Drugs in the original manufacturer packaging (except recalls)
- Undispensed pharmaceutical waste
- Unexpired waste or less than 1 year past expiration date
- Non-Creditable Waste:
- Broken or leaking container
- Repackaged
- Dispensed
- Expired (greater than 1 year past expiration date)
- Investigational new drugs
- Contaminated PPE
- Floor Sweepings
- Clean-up Material
- Evaluated (“Creditable”) Waste:
- Manufacturer credit already determined/verified
- Pharmaceuticals destined for TSDF
Non-creditable waste will no longer be able to flow through a Reverse Distributor. It must be disposed of by a permitted waste facility.
The flow chart below can help your healthcare facility determine the proper disposal method for its pharmaceutical waste.
6. Shipping Evaluated Vs. Non-Creditable Hazardous Pharmaceutical Waste
For evaluated and potentially creditable hazardous waste pharmaceuticals, the reverse distributor is exempt from RCRA Rules. Follow your reverse distributor’s shipping requirements. You must receive confirmation of delivery from the distributor.
Healthcare facilities need to have a manifest when shipping non-creditable hazardous waste pharmaceuticals. The good news is, as part of the August 21st changes, this process will become a little easier. Rather than having to write various hazardous waste codes on manifests, you will only need to write the word “PHARMS.” Remember though, that if the receiving state’s treatment facility has not adopted the rule, waste codes will still be required on the manifest. You’ll need to follow your facility’s internal procedures for collection containers and DOT Regulations when shipping the waste.
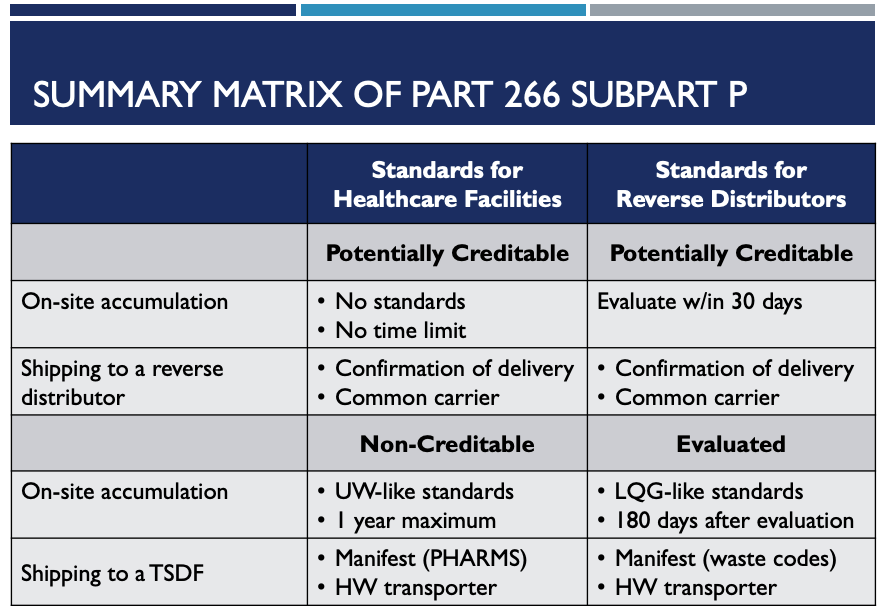
Summing Up 266 Subpart P
The following matrix will help both healthcare facilities and reverse distributors. It summarizes the timing for on-site accumulation and shipping standards for potential creditable pharmaceuticals.
Now that You Know…
Iowa healthcare facilities, we get it. It’s a lot to wrap your head around. Understanding the key changes above is a good start in avoiding harsh penalties after the new regulations go into effect August 21, 2019. Now that you’ve had a chance to get an overview, you have to do one more thing to do to demonstrate your knowledge….
Create a One Time Report
To let the EPA know you will be operating under Subpart P, it is requiring the submission of a one-time notification using this site EPA ID form: 8700-12.
Large Quantity Generators must submit this form during normal biennial reporting cycle. For Iowa and Alaska, this means the form is due March 1, 2020.
Small Quantity Generators need to complete this form within 60 days of the rule going into effect. For Iowa, that means the form is due Oct. 20, 2019.
Very Small Quantity Generators must complete form 8700-12 only if they decide to opt into Subpart P. There is no timeline for submittal.
While it’s a lot to take in, these changes clarify hazardous waste pharmaceutical disposal for healthcare facilities. And, help them be a better friend to the environment.
As always, if you have any questions, give us a call at 608-257-7652 or contact us at meriinc.com.
###